Difference between revisions of "Diels-Alder Reaction Experimental Design"
(→Optimum Experimental Design Problem) |
|||
| Line 58: | Line 58: | ||
<math> | <math> | ||
\begin{array}{cll} | \begin{array}{cll} | ||
| − | \displaystyle \min_{x, G, F, u} && trace(F^{-1} ( | + | \displaystyle \min_{x, G, F, u} && trace(F^{-1} (t__{end})) \\[1.5ex] |
\mbox{s.t.} \\ | \mbox{s.t.} \\ | ||
\dot{x}(t) & = & f(x(t), u(t),p), \\ | \dot{x}(t) & = & f(x(t), u(t),p), \\ | ||
| Line 201: | Line 201: | ||
|[20.0,100.0] | |[20.0,100.0] | ||
|} | |} | ||
| − | |||
== References == | == References == | ||
Revision as of 14:30, 4 December 2015
The Diels-Alder Reaction is an organic chemical reaction. A conjugated diene and a substituted alkene react and form a substituted cyclohexene system.
More information about the reaction can be found in ...
Model Formulation
Differential equation system:
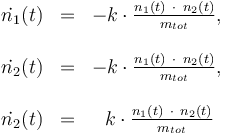
Solvent:
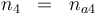
Reaction velocity constant:
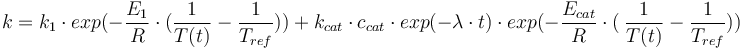
Total mass:
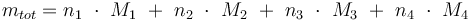
Temperature in Kelvin:
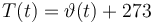
The ODE system is summarized to:
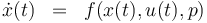
Optimum Experimental Design Problem
Failed to parse (syntax error): \begin{array}{cll} \displaystyle \min_{x, G, F, u} && trace(F^{-1} (t__{end})) \\[1.5ex] \mbox{s.t.} \\ \dot{x}(t) & = & f(x(t), u(t),p), \\ \\ \dot{h}(t) & = & \frac{n_3(t) \ \cdot \ M_3}{m_{tot}} \ \cdot \ 100 \\ \\ \dot{G}(t) & = & f_x(x(t),u(t),p)G(t) \ + \ f_p(x(t),u(t),p) \\ \\ \dot{F}(t) & = & w(t) (h_x(x(t),u(t),p)G(t))^T (h_x(x(t),u(t),p)G(t)) \\ \\ 0.1 & \le & n_{a1} \ \cdot \ M_1 \ + \ n_{a2} \ \cdot \ M_2 \ + \ n_{a4} \ \cdot \ M_4 \\ \\ 10 & \ge & n_{a1} \ \cdot \ M_1 \ + \ n_{a2} \ \cdot \ M_2 \ + \ n_{a4} \ \cdot \ M_4 \\ \\ 0.1 & \le & \frac{ n_{a1} \ \cdot \ M_1 \ + \ n_{a2} \ \cdot \ M_2 }{ n_{a1} \ \cdot \ M_1 \ + \ n_{a2} \ \cdot \ M_2 \ + \ n_{a4} \ \cdot \ M_4 } \\ \\ 0.7 & \ge & \frac{ n_{a1} \ \cdot \ M_1 \ + \ n_{a2} \ \cdot \ M_2 }{ n_{a1} \ \cdot \ M_1 \ + \ n_{a2} \ \cdot \ M_2 \ + \ n_{a4} \ \cdot \ M_4 } \\ \\ 0 & = & \vartheta_{lo}, \forall \, t \in [t_0,2] \\ \\ 0 & = & \vartheta_{lo} + \frac{t-2}{6} ( \vartheta_{up} - \vartheta_{lo} ) , \forall \, t \in [2,8] \\ \\ 0 & = & \vartheta_{up}, \forall \, t \in [8,t_{end}] \\ \\ x & \in & \mathcal{X},\,u \in \mathcal{U},\, p \in P. \end{array}
| Name | Symbol | Initial value ( ) )
|
| Molar number 1 | 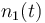
|
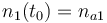
|
| Molar number 2 | 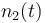
|
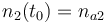
|
| Molar number 3 | 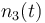
|
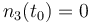
|
| Name | Symbol | Value |
| Molar Mass | 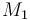
|
0.1362 |
| Molar Mass | 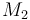
|
0.09806 |
| Molar Mass | 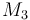
|
0.23426 |
| Molar Mass | 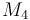
|
0.236 |
| Universal gas constant | 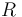
|
8.314 |
| Reference temperature | 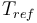
|
293 |
| St.dev of measurement error | 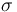
|
1 |
| Name | Symbol | Value |
| Steric factor | 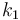
|
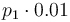
|
| Steric factor | 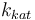
|
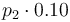
|
| Activation energie | 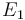
|
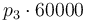
|
| Activation energie | 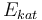
|
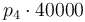
|
| Catalyst deactivation coefficient | 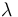
|
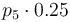
|
with 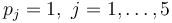
| Name | Symbol | Interval |
| Initial molar number 1 | 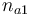
|
[0.4,9.0] |
| Initial molar number 2 | 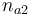
|
[0.4,9.0] |
| Initial molar number 4 | 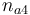
|
[0.4,9.0] |
| Concentration of the catalyst | 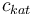
|
[0.0,6.0] |
| Initial molar number 1 | 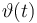
|
[20.0,100.0] |
References
R. T. Morrison and R.N. Boyd. Organic Chemistry. Allyn and Bacon, Inc., 4th edition, 1983 \\ Dissertation Stefan Körkel